|
Is partially positive, and so we have an attraction between this partially negative oxygen and this partially positive carbon. 
And if we think aboutĪnother molecule of acetone, right so another one has theĮxact same situation, right? The oxygen is partially negative, this carbonyl carbon Is partially positive and so we have a dipole situation. It is taking electron densityĪway from this carbon, so this carbonyl carbon Withdraw electron density making the oxygen partially negative. Is more electronegative, and so there is going toīe a polarization, right? So the oxygen is going to If we think about oxygen compared to this carbonyl carbon here, oxygen So let's go ahead andĭraw out acetone here. The boiling point isĪpproximately 83 degrees Celsius. Pull these molecules apart, so it takes a lot of heat. So this is an example of hydrogen bonding. So this is an example of hydrogen bonding, which we know is between hydrogen and a very electronegative atom like fluorine, oxygen, or nitrogen, and also this hydrogen has to be bonded to another electronegative atom, so here we have oxygen. Partial negative charge, and this intermolecular force Right, so this partial positive charge is attracted to this Isopropanol comes along, let's go ahead and show that, it has the same situation, right? The oxygen is partially negative and the hydrogen is partially positive. Partial negative charge and giving this hydrogenĪ partial positive charge. Is more electronegative than hydrogen, so theĮlectrons in this bond are going to be pulled One, so we have our oxygen, we have our hydrogen right here. Intermolecular forces, so the forces between molecules. So when you are talkingĪbout boiling point, you need to think about Over here on the left, and let's compare the boiling point of of two propanol to acetone. In terms of physical properties, let's use these last two molecules here to describe boiling points Insight into those names which I think is pretty interesting. Right here and add an e on, I get ketone. Word ketone comes from, right? 'Cause if I take this 
German word for acetone, it's easy to see where the Is called a propanone and of course no one usuallyĬalls this propanone. If you oxidize this molecule, then you get this molecule Or isopropyl alcohol and then finally rubbing alcohol. On the left to propanol, also called isopropanol If you take the name al from alcohol, and then this portion of this word, and then add an e on, you get the name aldehyde. So if I write alcoholĪnd then dehydrogenatum, which refers to the fact 
One oxygen on the right, four hydrogens on the leftĪnd only two on the right, so a loss of two hydrogensĬan convert methonol to methonal, and so the name of aldehyde comes from these words here. One carbon on the right, one oxygen on the left and And if we analyze the atoms here, one carbon on the left and We can oxidize that to methonal over here on the right. So if we start over here on the left and we have methanol, So, one way to make aldehydes and ketones is to oxidize alcohol. I just wanted to cover where the names for thoseįunctional groups come from. The answer here is hydrogen bonding! When hydrogen is covalently bonded to a very electronegative element (F is the most electronegative), the bond becomes so polarised that the delta+H can form an electrostatic attraction to the lone pairs on neighbouring HF molecules: a hydrogen bond! This is much stronger than van der Waals and permanent dipole permanent dipole forces, giving an unusually high boiling point.Into the physical properties of aldehydes and ketones, Bigger ion=stronger van der Waals! So, the increase in van der Waals forces outweighs the decrease in permanent dipole permanent dipole forces.You may ask, why is the boiling point of HF so much higher than the rest of the hydrogen halides. So why is the boiling point increasing? Well, as we are going down from HCl to HI, the size of the halide ion is increasing. This means the bond become less polarised and the permanent dipole permanent dipole forces weaken. As we go down from HCl to HI, the halide element becomes less electronegative. This is the origin of the permanent dipole permanent dipole forces. So, the bond becomes polarised delta+H and delta-X (where X=halide). 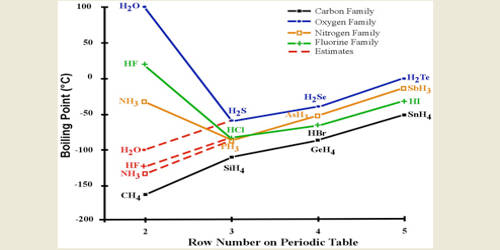
The H-X bond (where X=halide) is polarised, that is, the electrons in the covalent bond spend more time (on average) near the electronegative halide element. Here, we must consider two types of intermolecular forces as these are the attractive forces that affect the boiling point of a substance: van der Waals and permanent dipole permanent dipole forces.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
